Botulinum neurotoxin for sialorrhea: a first choice therapy
Introduction
Botulinum neurotoxin (BoNT) has been approved as a safe, minimal invasive procedure and therapeutic option for several interdisciplinary indications in the head and neck region in the last decades. In 1979, Scott first used BoNT in strabismus correction (1). The powerful poison that was responsible for the clinical appearance of botulism turned out to be a useful and effective therapeutic tool. The expansion of its use in functional disorders especially of the head and neck region was enormous. The toxins efficacy is based on its specific, but reversible inhibition of neurotransmitter (acetylcholine) release in synapses (2). This condition is not only relevant in the management and reduction of involuntary muscular contractions and pathological movements. In 1994, the pioneer in BoNT therapies for functional disorders Laskawi, first described the possible extension of BoNT use in autonomic disorders, reporting that the toxin could block the release of acetylcholine as it did in muscles and further the cholinergic parasympathetic secromotor fibers in salivary and sweat glands, too (3). This medical involution resulted a new era in the treatment of hypersalivation, hyperhidrosis and other secretory situations such as Frey’s syndrome and rhinitis (allergic and vasomotor) (Table 1).
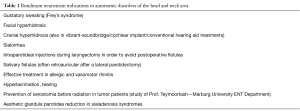
Full table
In a healthy adult the daily salivary production is approximately 0.75 to 1.5 litres, while the submandibular gland secretes around 50% of the total saliva, the parotid gland contributes 40% and the sublingual glands about 5% (4,5). Minor salivary glands of the oral cavity and the pharynx produce the rest. The stimulation of the salivary glands arises mainly from the parasympathetic nervous system over the ganglion oticum (parotid gland) or the ganglion submandibular (submandibular and sublingual glands) and leads to acetylcholine release onto the salivary acinar cells. This transmitter binds to muscarinic receptors and causes an increased calcium ion activity intracellular that induces secretion (5). Sympathetic muscular contraction enhances the expression of saliva after its stimulation (6).
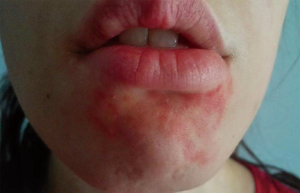
Sialorrhea is defined as the involuntarily outcome of saliva beyond the lip margin, a normal phenomenon during childhood, which is considered to be pathologic when it occurs beyond the fourth year of life (7). This excessive saliva production and incontinence of remaining within the mouth can contribute to drooling (extensive Hypersalivation) (Figure 1). Disorders that are associated with this serious symptomatology include neurologic entities as cerebral palsy, stroke, Morbus Parkinson, amyotrophic lateral sclerosis, etc. Deficits with influence on the body and jaw posture, the perioral muscular system control and the mental situation can often lead to an uncontrolled saliva flow (8). Further, dysphagia, odynophagia, an upper esophagus sphincter stenosis or special anatomic conditions of the stomatopharynx (tongue size, tonsil size, mandibular occlusion) or the upper aerodigestive tract (stenosis, surgical changes) can cause hypersalivation. The observation that saliva can have a negative effect on wound healing processes by inducing even fistulas after laryngectomy, led also to the idea of a temporary reduction of the salivary glands production by performing BoNT injections intraoperative (9).
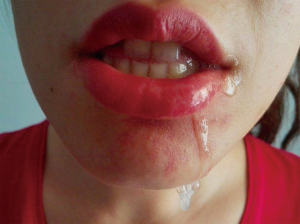
Administration methodology
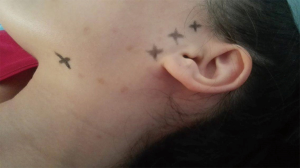
The primary modality in the conservative sialorrhea treatment is BoNT. Injections may be performed using a blind palpation of the glands, sonographic or EMG guidance. In our practice, the glandula submandibularis is treated sonography-guided in a single injection of 15 units Botox on each side. A three point technique with distribution of 7.5 units Botox per injection-point is been performed for each glandula parotidea separately (Figure 2). When injecting with ultrasound assistance, the needle tip is not always visualized. The intraglandular parenchyma motion while the substance is being injected gets clearly visible and allows to estimate the depth of the tissue that is being treated. Although BoNT can be injected only in the parotid gland or solely the submandibular gland, we have noted that the sialorrhea controlling was even better when both gland types where treated (submandibular and parotid). The risk of toxin diffusion to local musculature is low if the treated area is within the gland and as long as BoNT is injected slowly into the gland. Superficial anesthesia has never been required in our patients. The use of 30 G needles allow comfortable, well tolerated injections.
Discussion
Various noninvasive and more aggressive techniques have been practiced in the last decades in order to control pathologic sialorrhea symptoms. The most common approach is to prescribe anticholinergic medications like scopolamine and glycopyrrolate that work by blocking the parasympathetic nervous system and innervation of the salivary production. Despite the fact that these pharmaca groups have many contraindications (per example gastrointestinal motility disorders, glaucoma), over 30% of patients undergoing such a treatment are unable to tolerate the medications, due to significant side-effects that have been described (behavioral changes, severe constipation etc.) (10). Radiation of the salivary glands is another curative option for sialorrhea patients. The malignancy potential of this alternative transforms it into a therapy reserved only for special cases of elderly patients (11). Several invasive interventions such as a tympanic neurectomy to denervate the salivary glands, a major salivary gland relocation to reroute the saliva flow with the risk of aspiration and surgical excisions of the main salivary glands have been performed to improve sialorrhea. Transtympanic neurectomy at the tympanic plexus is a reversible option, due to nerve fiber regeneration that occurs in less than 18 months postoperative (12). Shott et al. described the bilateral submandibulectomy combined even with parotid gland ligation or rerouting as highly effective but aggressive method, entailing the possibility of a ramus marginalis weakness of the mandibular nerve and external incisions that are necessary (13). All these techniques can be effective, but they have more disadvantages than benefits for the patients in comparison to the noninvasive neurotoxin injections. Beyond the effectiveness of BoNT in glandular secretions it is reported to have a remarkable longer duration in autonomic neurons as on muscles. BoNT has a longer-lasting positive effect from six up to twelve months in hyperhidrosis or sialorrhea, compared to the three to four months observed in muscular tone treatments (14-16).
Sialorrhea has been associated with complications such as dehydration or frequent dermal infections or even local ulcera as it affects the perioral skin area (Figure 3). In some clinical conditions of reduced laryngeal sensitivity, saliva may reach the tracheobronchial system leading to an aspiration pneumonia. Excess salivation can challenge our cultural definitions of social standing, as it stigmatizes the patients, leading often to a negative influence in their quality of life and in some cases even to a social isolation. Based on our experience as well as those of other groups, we recommend the use of intraglandular BoNT injections in sialorrhea patients as an efficacious treatment that combines safety and a long duration of results. The high side-effect profile of anticholinergic medications limits their usage in clinical practice and gives space to the catholic usage of BoNT as a first line treatment option for hypersalivation.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Scott AB. Botulinum toxin injection into extraocular muscles as an alternative to strabismus surgery. Ophthalmology 1980;87:1044-9. [Crossref] [PubMed]
- Heckmann M, Rzany B. Botulinumtoxin in der Dermatologie, Grundlagen und praktische Anwendung; Anatomie der mimischen Muskulatur; Urban&Vogel, München, 2006.
- Drobik C, Laskawi R. Frey-Syndrom: Behandlung mit Botulinum-Toxin. HNO Aktuell 1994;2:142-4.
- Dawes C. Circadian rhythms in human salivary flow rate and composition. J Physiol 1972;220:529-45. [Crossref] [PubMed]
- Strutz J, Mann W. Praxis der HNO-Heilkunde, Kopf und Halschirurgie 2. Auflage, Georg Thieme Verlag, 2010:98.
- Blitzer A, Benson BE, Guss J. editors. Botulinum Neurotoxin for Head and Neck Disorders. New York: Thieme Medical Publishers, 2012:212.
- Bavikatte G, Sit PL, Hassoon A. Management of drooling of saliva. BJMP 2012;5:a507.
- Stuchell RN, Mandel ID. Salivary gland dysfunction and swallowing disorders. Otolaryngol Clin North Am 1988;21:649-61. [PubMed]
- Ihler F, Laskawi R, Matthias C, et al. Einsatz von Botulinum Toxin A nach mikrovaskulären ALT-Lappen bei einem Patienten mit Zungen- uns Mundbodenkarzinom. HNO 2012;60:905. [Crossref]
- Mier RJ, Bachrach SJ, Lakin RC, et al. Treatment of sialorrhea with glycopyrrolate: A double-blind, dose-ranging study. Arch Pediatr Adolesc Med 2000;154:1214-8. [Crossref] [PubMed]
- Borg M, Hirst F. The role of radiation therapy in the management of sialorrhea. Int J Radiat Oncol Biol Phys 1998;41:1113-9. [Crossref] [PubMed]
- Frederick FJ, Stewart IF. Effectiveness of transtympanic neurectomy in management of sialorrhea occurring in mentally retarded patients. J Otolaryngol 1982;11:289-92. [PubMed]
- Shott SR, Myer CM 3rd, Cotton RT. Surgical management of sialorrhea. Otolaryngol Head Neck Surg 1989;101:47-50. [PubMed]
- Ellies M, Gottstein U, Rohrbach-Volland S, et al. Reduction of salivary flow with botulinum toxin: extended report on 33 patients with drooling, salivary fistulas, and sialadenitis. Laryngoscope 2004;114:1856-60. [Crossref] [PubMed]
- McGeachan AJ, Mcdermott CJ. Management of oral secretions in neurological disease. Pract Neurol 2017. [Epub ahead of print]. [Crossref] [PubMed]
- Barbero P, Busso M, Artusi CA, et al. Ultrasound-guided Botulinum Toxin-A Injections: A Method of Treating Sialorrhea. J Vis Exp 2016;(117).
Cite this article as: Karapantzou C, Karapantzos I, Zarogoulidis P, Karanikas M, Thomaidis V, Charalampidis C. Botulinum neurotoxin for sialorrhea: a first choice therapy. Ann Res Hosp 2017;1:17.